Immunity Module – Molecular Medicine
Question 1: Flow Cytometry
The gating strategy used in this case is forward and side scatter gating. This form of gating is relatively generous and is aimed at identifying all cells of interest. The strategy differentiates cells based on their differences in sizes and complexity (Givan, 2013). Also, it attempts to eliminate debris that may react to treatments randomly hence giving no predictable results. The goal of eliminating debris in the forward and side scatter gating is because they are not of interest. When this gating technique is used, major densities are captured. However, the technique ignores events with very low FSC and SSC, as well as the densities with very high FSC and SSC (Wong & Davis, 2013). Analysis of the diagrams provided in the case indicate that the plots of FSC vs. SSC were made. In addition, it is evident that the researcher attempted to include as many cell densities within the gates as possible. This implies that the goal was to include all regions of activity to facilitate the subsequent identification of the active cell types based on the FMO control. In addition, the gating technique ignores the events with very low SSC and FSC as noted in both untreated controls and under the thioglycollate treatments.
Fluorescence Minus One (FMO) control is adopted to build a multicolored cytometric panels. The approach dictates the setting up of gates to facilitate the acquisition of data on the various fluorophores that are brightly noticeable. The technique involves setting up the gates such that all fluorophores are included within the gate except one fluorophore at any given time (Kőszegi, 2016). As a result, it is possible to determine the fluorescent properties lost as a result of the excluded fluorophore. Critical analysis of the FMO control used in the case indicates that priciples on fluorophore exclusion are not respected. For instance, the first case subjected to UV analysis did not exclude any fluorophore. The second control under Blue 660/20 excluded all the fluorophores. The control under Blue 575/26 partially excludes a fluorophore while the last case almost excluded all fluorophores.


Graph and Inhibitor Choice
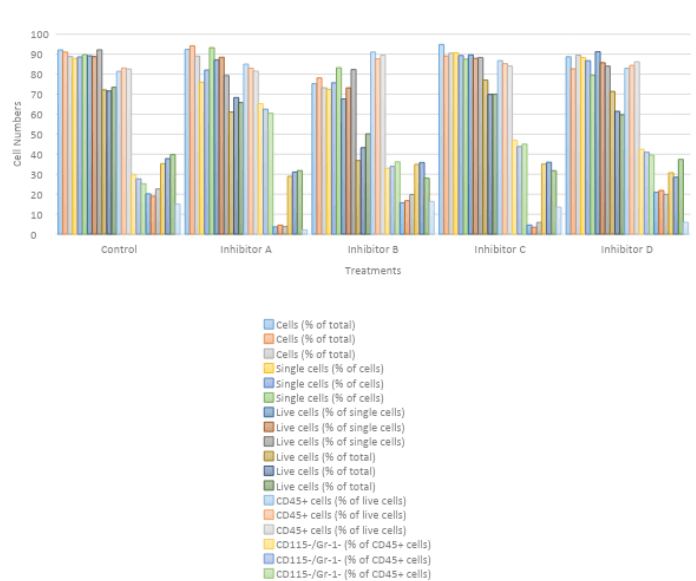
Based on the data, the best compound to use would be inhibitor A. the choice of this compound is informed by its impacts on the numbers CD115-/Gr-1+. Studies indicate that these cells are involved in early inflammatory bursts and are thus key in the subsequent recruitment of neutrophils at the site of inflammation.
Assays for Neutrophil Inhibition
There are multiple assays that can be conducted to determine the mechanisms of neutrophil inhibition. The first assay that should be considered is cell count. This approach will help to determine the changes in the relative cell numbers hence establish the cells that are likely to have played a role in the inhibition (Spisani et al., 2001). Random locomotion assays can then be initiated to evaluate the movement of neutrophils in different compartments and across different pore sizes. This assay can be done in microchemotactic chambers which also involves the estimation of distances moved by the cells in a 90 minutes window. Chemotactic analysis is also conducted to determine whether the inhibitor triggers generation of chemical compounds that affect the migration of the neutrophils to the site of action. Moreover, superoxide anion production can be assayed, with the concentration of superoxide dismutase in the bloodstream being a key determinant of the enzyme titers (Spisani et al., 2001). The estimation of superoxide anion may also involve analysis of wavelengths to determine the changes in absorbance values which could be reflective of the neutrophil activity in the given tissue.

In addition, the researchers could conduct enzyme assays, with lysozyme activity being an important factor. Lysozyme is often found as part of the neutrophil granule enzymes and is key in the degradation and killing of external cells. Inhibitors that act through degranulation are likely to inactivate the lysozyme enzymes as a way of crippling the activities of the neutrophils. The process of assaying for the degranulation process can be done by incubating them in cytochalasin B for 10 minutes. The other technique for assaying for the techniques involved in the inhibition of neutrophil recruitment is the assay for changes in calcium ion concentration. The activity of neutrophils is directly influenced by the concentrations of the calcium ions. Therefore, inhibition of neutrophils could occur through alteration of calcium ion concentrations in the tissues. The other approaches that can be used to determine the mode of inhibition of neutrophil recruitment include cell viability tests, receptor binding tests and treatments with CsA (Spisani et al., 2001). Cell viability tests focus on the impacts of the tested drugs on the cells while the receptor binding experiments focus on the activation mechanisms of the neutrophils.
Question 2: Vaccine Development
The case indicates that mutations in the gene coding for the bacterial proteins has resulted into the loss of efficacy of the original vaccine. Therefore, the new vaccine should be able to target the specific differences between the bacterial and human proteins. The key considerations that must be made in the development of the new vaccine include the antigens to be targeted, adjuvants to be used, vaccine delivery systems and the technical hurdles that might be faced. Assuming there are no technical and financial constraints, then the recommended approach would involve two stages; the pre-clinical and the clinical stages.
In the pre-clinical stage, focus should be directed on the identification of the alternative target antigens. This would require critical analysis of the differences between the bacterial FcσRII and the human FcβRI proteins. After the identification of the differences, antigen testing should be carried out for the different regions. Subsequently, the vaccine concept should be developed for the specific bacteria. This should then be followed by the laboratory analysis of the vaccine concept to determine its viability. Viable preliminary vaccines are then tested in animals such as guinea pigs before eventually undergoing the three phases of clinical trials. Successful vaccines can then be used to vaccinate susceptible populations against Clostridium tympani, with constant post-marketing analyses conducted to ensure that there are no adverse effects.



Vaccine efficacy is considered as the percentage reduction in the disease prevalence in the vaccinated population compared to the unvaccinated population, assuming the most favorable environments (Buckland, 2005). It can thus be a powerful tool for predicting the relative risk of different individuals in defined population to the attacks by Clostridium tympani. The efficacy of the vaccine for this bacterium can be determined during the clinical and post-clinical trials. Following the development of the vaccine, it is administered to a population of individuals who are predisposed to attacks by the bacterium. On the other hand, a second population is used as a control group and is thus not given the vaccine treatment. Since the control and the study groups are within the same environments, their environmental predisposal to the bacterial attack is similar. As such, any notable differences in disease incidences between the two populations can be deemed as results of the vaccine treatment.
Individuals are randomly selected from the treated group and the control group, and blood samples drawn for assay to determine whether or not they have the bacteria (Buckland, 2005). The number of samples from each group recording cases of the bacterial infections are recorded and the difference in prevalence determined. The next step is to compute the ratio of the difference in infection to the number of the unvaccinated cases of infection, a value which is then converted into a percentage to determine the percentage efficacy of the vaccine. Alternatively, the test for the efficacy of the vaccine can be done at a pre-clinical phase whereby a population of guinea pigs are treated with the vaccine while another population is left untreated. The two groups are then exposed to the bacterial antigen and the rates of infection determined. Any observed differences in infection rates as determined through differences in antibody concentrations are considered to indicate efficacy of the vaccine.
The preferred adjuvant for use in this case is Montanide adjuvant which also works on the principle of water-in-oil emulsion. The primary benefit of Montanide is that it can support diverse types of antigens, including those that are unique and difficult to obtain. In addition, weakly immunogenic antigens as well as those of low molecular weight can be effectively supported. However, Montanide adjuvant does not have the toxic effects observed in Freund’s adjuvant (Klimka et al., 2015). Its injection into the body does not trigger local inflammations as observed in Freund’s adjuvant. However, the use of this adjuvant has been relatively limited to veterinary medicine.
Question 3: Polysaccharide Conjugate Vaccines
Graph of Opsonized Phagocytosis Assay
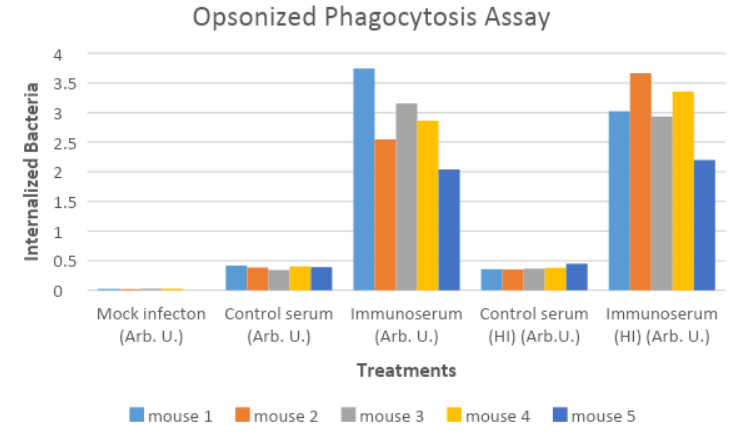
The graphs indicate relatively low titers in mock infections and in the control serums. In the mock infections, the absence of the antigens are result in relatively low rates of opsonization hence the number of macrophages that have phagocytosized bacteria is negligible. In the control groups, the rate of phagocytosis is also low since the bacterial populations are low. However, the immunized serum reflected higher rates of phagocytosis in all mice both when subjected to heat treatment or not. This shows that there is negligible effect of the heat treatment on the efficacy of the vaccine. Since protein components are often denatured at high temperatures, the absence of significant change in the activity of the vaccine indicates that the polysaccharide component unit is involved in receptor binding hence the identification of the bacteria (Zangeneh, Baracco & Al-Tawfiq, 2011).
Macrophages have specific surface properties that can be used to determine their activation statuses. Under conditions of health, macrophages are in resting state hence express normal surface proteins. The receptors are unoccupied and their abundance on the cell surfaces relatively low (Murphy et al., 2010). In cases of infection, however, there is activation of the macrophages to express specific receptors on the surface. In the case of N. mengitides infection, the macrophages become activated to express specific surface receptors. These may include polysaccharide conjugate receptors that bind to the antigens on the surface of the bacteria to facilitate their recognition and subsequent phagocytosis. Therefore, activated macrophages can be identified based on the surface molecules expressed. Besides, macrophages are involved in the release of certain pro-inflammatory cytokines. An analysis of the patterns of cytokine concentrations in serum can be used to determine the activation state of the macrophages. In cases of high concentrations of the pro-inflammatory cytokines, it can be deduced that the macrophages are highly activated by the bacteria (Murphy et al., 2010). On the contrary, low concentrations of the cytokines could suggest limited activation of the macrophages.
1. Buckland, B. (2005). The process development challenge for a new vaccine. Nature Medicine, 11(4s), S16-S19. http://dx.doi.org/10.1038/nm1218
2. Givan, A. (2013). Flow Cytometry (2nd ed.). Somerset: Wiley.
3. Klimka, A., Michels, L., Glowalla, E., Tosetti, B., Krönke, M., & Krut, O. (2015). Montanide ISA 71 VG is Advantageous to Freund’s Adjuvant in Immunization AgainstS. aureusInfection of
4. Mice. Scandinavian Journal Of Immunology, 81(5), 291-297. http://dx.doi.org/10.1111/sji.12279
5. Kőszegi, T. (2016). Flow cytometry in rapid mycotoxin testing. Cytometry Part A, 91(2), 173-174. http://dx.doi.org/10.1002/cyto.a.22939
6. Murphy, B., Bush, H., Sundareshan, V., Davis, C., Hagadone, J., & Cory, T. et al. (2010). Characterization of macrophage activation states in patients with cystic fibrosis. Journal Of Cystic
7. Fibrosis, 9(5), 314-322. http://dx.doi.org/10.1016/j.jcf.2010.04.006
8. Spisani, S., Fabbri, E., Muccinelli, M., Cariani, A., Barbin, L., Trotta, F., & Dovigo, L. (2001). Inhibition of neutrophil responses by cyclosporin A. An insight into molecular mechanisms.
9. Rheumatology, 40(7), 794-800. http://dx.doi.org/10.1093/rheumatology/40.7.794
10. Wong, L., & Davis, B. (2013). Monochromatic gating method by flow cytometry for high purity monocyte analysis. Cytometry Part B: Clinical Cytometry, 84B(2), 119-124. http://dx.doi.org/10.1002/cyto.b.21053
11. Zangeneh, T., Baracco, G., & Al-Tawfiq, J. (2011). Impact of conjugate pneumococcal vaccines on the changing epidemiology of pneumococcal infections. Expert Review Of Vaccines, 10(3), 345-353. http://dx.doi.org/10.1586/erv.11.1



The download will start shortly.

The download will start shortly.
 Subject:
Medicine
Subject:
Medicine  Number of pages: 3
Number of pages: 3  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 9
Number of pages: 9  Subject:
Medicine
Subject:
Medicine  Number of pages: 2
Number of pages: 2  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 11
Number of pages: 11  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 9
Number of pages: 9  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 13
Number of pages: 13  Subject:
Medicine
Subject:
Medicine  Number of pages: 46
Number of pages: 46  Subject:
Nursing
Subject:
Nursing  Number of pages: 4
Number of pages: 4  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 10
Number of pages: 10  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 10
Number of pages: 10  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 6
Number of pages: 6  Subject:
Nursing
Subject:
Nursing  Number of pages: 2
Number of pages: 2  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 9
Number of pages: 9  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 1
Number of pages: 1  Subject:
Health and Social Care
Subject:
Health and Social Care  Number of pages: 2
Number of pages: 2 
